 When heated, it melts and the crystallinity is lost. These intermolecular forces are even stronger when the polymer chains pack together in a regular way as in HPDE (high density poly(ethene)) and have regions of crystallinity. Their larger sizes lead to much stronger intermolecular forces leading in turn to much higher melting points, and the characteristic properties of hardness and flexibility. The major differences between smaller molecules and polymers lie not with their chemical properties but with their physical ones. If it contains an aromatic ring, as in poly(phenylethene) (often known as polystyrene), it will undergo substitution reactions, say with nitric acid. For example, if the polymer contains a carbon-carbon double bond, as in poly(but-1,3-diene), it will undergo additions reactions with, say hydrogen or bromine. Their chemical properties are similar to those of simple molecules. Polymers are large molecules, a type of macromolecule. Finally the unit summarises the range of processing techniques that can be used to convert polymers into a vast range of different products.Įach of the other units in the Polymers section describes the manufacture, properties and uses of an individual polymer or group of polymers in more detail. 
This unit provides an overview of the main types of polymers characterised by how they are made, how their structures govern their general properties and how these properties can be refined by their formulation using a range of additives. The polymer is a chain of atoms, providing a backbone, to which atoms or groups of atoms are joined. The simple compounds whose molecules join together to form the polymers are called monomers. Dehydration reactions typically require an investment of energy for new bond formation, while hydrolysis reactions typically release energy by breaking bonds.When many molecules of a simple compound join together, the product is termed a polymer and the process polymerization. Dehydration and hydrolysis reactions are similar for all macromolecules, but each monomer and polymer reaction is specific to its class. When polymers are broken down into smaller units (monomers), a molecule of water is used for each bond broken by these reactions such reactions are known as hydrolysis reactions. These types of reactions are known as dehydration or condensation reactions. A monomer joins with another monomer with the release of a water molecule, leading to the formation of a covalent bond. The polymer is more than the sum of its parts: it acquires new characteristics, and leads to an osmotic pressure that is much lower than that formed by its ingredients this is an important advantage in the maintenance of cellular osmotic conditions. Macromolecules are made up of single units known as monomers that are joined by covalent bonds to form larger polymers. Proteins, carbohydrates, nucleic acids, and lipids are the four major classes of biological macromolecules-large molecules necessary for life that are built from smaller organic molecules. 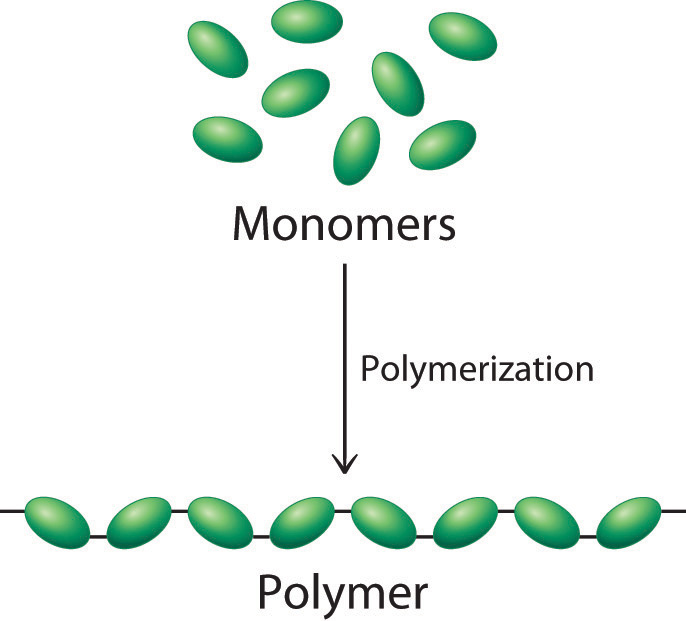
Breakdown of these macromolecules provides energy for cellular activities. Proteins are broken down by the enzymes pepsin and peptidase, and by hydrochloric acid. 
For instance, carbohydrates are broken down by amylase, sucrase, lactase, or maltase. Each macromolecule is broken down by a specific enzyme. This allows for easy absorption of nutrients by cells in the intestine. For example, in our bodies, food is hydrolyzed, or broken down, into smaller molecules by catalytic enzymes in the digestive system. These reactions are similar for most macromolecules, but each monomer and polymer reaction is specific for its class. \).ĭehydration and hydrolysis reactions are catalyzed, or “sped up,” by specific enzymes dehydration reactions involve the formation of new bonds, requiring energy, while hydrolysis reactions break bonds and release energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
